1.A spectrophotometer utilizes monochromatic light of specific wavelength provided by a monochromator or a special light source passing through standard sample and the analyzed sample to analyze the composition of a substance by comparing the light intensity of the two samples.
2.Common wavelength range: 200——400nm UV light region (UV spectrophotometer)、400——760nm visible region(visible spectrophotometer/ colorimeter) 、2.5——25μm IR region(IR spectrophotometer/ atomic absorption spectrophotometer)
3.The following instrument is an essential detecting appliance for food factory and drinking water plant applying for QS and HACCP certification.

The picture of spectrophotometer
The spectrophotometer adopts a light source that produces multiple wavelengths, and through a series of spectrophotometer, a specific wavelength light source is generated. After the light source passes through the tested sample, some light sources are absorbed. Calculate the absorption value of the sample and convert it into the concentration of the sample. The absorption value of the sample is proportional to the concentration of the sample.
The formula converted
A=-log(I/I。)=-lgT=KLC
Where A is the absorbance
I。is the intensity of incident monochromatic light
Iis the intensity of transmissive monochromatic light
is the transmittance of substance
K is the molar absorption coefficient
L is the optical path of the analytical substance, side length of the cuvette
C is the concentration of the substance
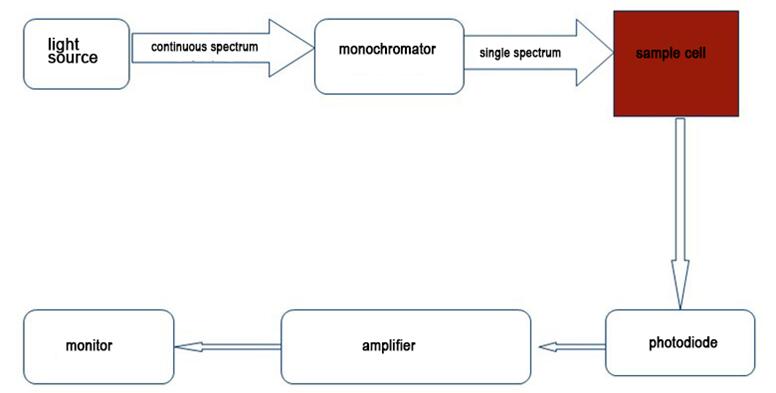
Basic principle structure diagram
Recommended model
UV spectrophotometer
Receiver:
UV-035DQ、 UV-035EQ、UV-035DQC、UV-035EQC
Visible spectrophotometer
Receiver:
PIN-13DSB
Light source
64258, 01312-U
RM 707-708, 258 Guoxia Road, Yangpu District,Shanghai, China PC:200433
Copyright © 2019-2020.Light-Catcher Co.,Ltd All rights reserved.沪ICP备15002270号-1

